|
Serum proteins in particular have been shown to alter the biological activity of EVs. These contaminants can lead to EV circulating DNA and long RNAs also precipitate under these conditions, making it difficult to assign a biological phenotype to any one molecular entity. Finally, use of crowding reagents, particularly on fluids such as serum/plasma, tend to co-precipitate large proteins and ribonucleic acid:protein complexes such as Ago-complexed miRNA. A separate first step isolation method is SEC systems, which allow for little more than 20 mL input. The role of extracellular vesicles (EVs) has received considerable attention in recent years, in particular the class of EVs with a diameter 100,000 g) onto EV, risking lysis and denaturation. From our results, we propose that cross-flow/Capto Core isolation is a robust method of purifying highly concentrated, homogenous, and functionally active EVs from industrial-scale input volumes with few contaminants relative to other methods. Moreover, EVs isolated through this method can be further concentrated down or directly immunopurified to obtain discreet populations of EVs. It outperforms other isolation procedures based on a number of biochemical and biophysical assays. Through this combination, EVs loss is kept to a minimum. 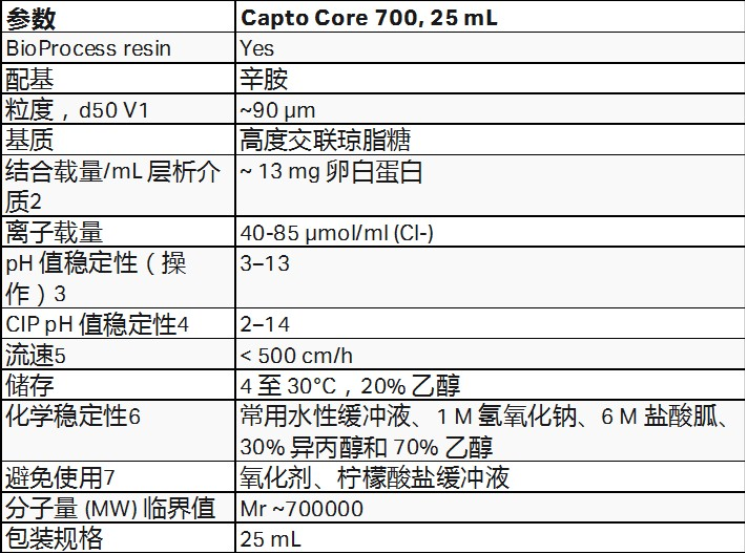  
Here, we present an industrial-scale workflow for isolating highly pure and functional EVs using cross-flow based filtration coupled with high-molecular weight Capto Core size exclusion. Common measures to quantify relative success are: concentration of the EVs, purity from non-EVs associated protein, size homogeneity and functionality of the final product. Isolation of extracellular vesicles (EVs) from cell culture supernatant or plasma can be accomplished in a variety of ways.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
